Personal Wealth Management / Market Analysis
Innovation Gives Health Care Stocks a Shot in the Arm
Drug innovation looks poised to continue boosting Health Care stocks.
After trailing in 2016 and 2017, global Health Care stocks are outperforming nicely this year—up 7.2% year to date, versus the MSCI World Index’s -1.4% decline—as investors are finally starting to appreciate their positive fundamentals.[i] While the sector—and particularly Pharmaceuticals—had plenty going for it in prior years, political fears over price controls and efforts to repeal the Affordable Care Act weighed on sentiment. More recently, investors fretted potential patent expirations and the president’s Twitter attacks on several Health Care companies. However, thus far, none of these fears have translated to actual policy, and firms have taken expiring patents in stride, allowing sentiment toward Health Care to start healing. As fears continue fading, we believe Health Care should continue benefiting from still underappreciated improvement—including new drug approvals and booming drug innovation.
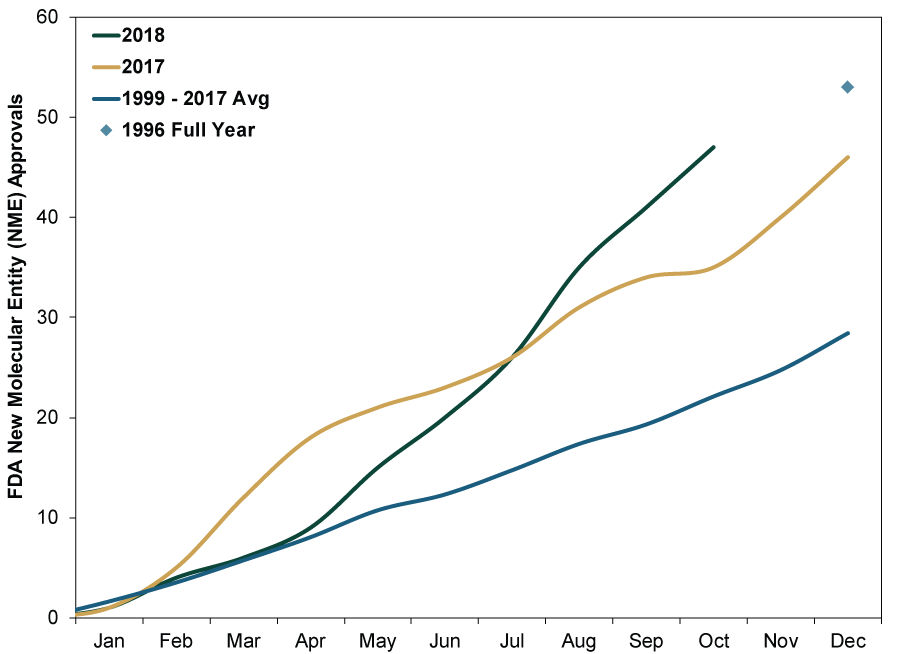
Thanks to a more favorable regulatory environment, 2018 drug approvals have been strong. The Food and Drug Administration (FDA) approved 47 new drugs in 2018’s first 10 months—more than doubling the full-year average since 1999 and on pace to break 1996’s record (53)—as shown in Exhibit 1, which tracks approval of New Molecular Entities, or drugs that weren’t previously approved (i.e., not generic versions of existing drugs). This sharp jump stems partly from a more streamlined drug-approval process. In December 2016, President Obama signed the 21st Century Cures Act, expanding funding to the National Institutes of Health for clinical research and liberalizing the types of clinical trials the FDA can accept as evidence of a drug’s efficacy. In short, the law simplifies drug approvals. In our view, this regulatory easing—a positive for Pharmaceuticals companies—is still widely underappreciated.
Exhibit 1: FDA Approvals for New Molecular Entities
Source: US Food and Drug Administration, as of 10/29/2018. New Molecular Entity (NME) approvals, 12/31/1996 – 10/31/2018.
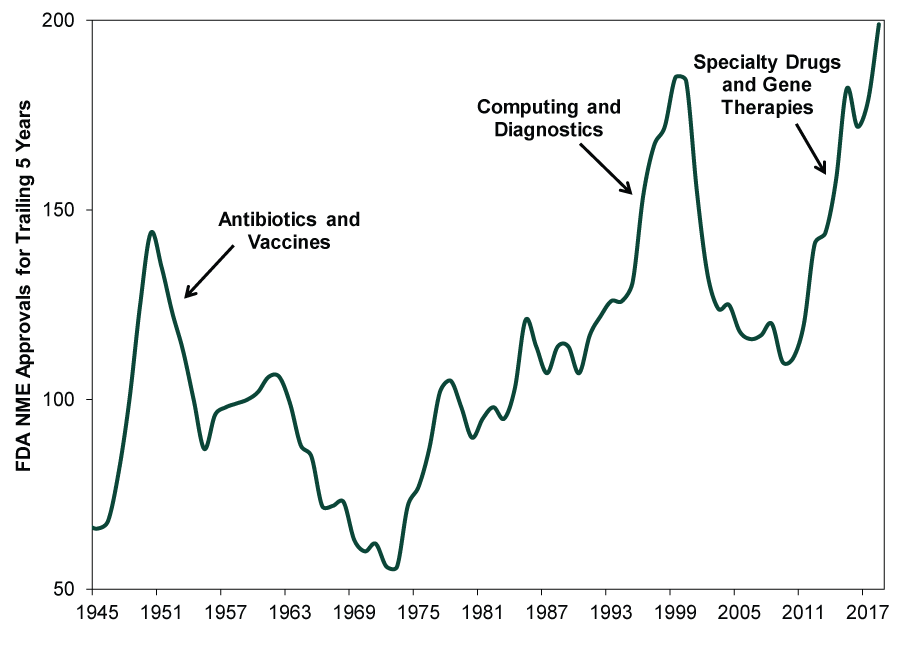
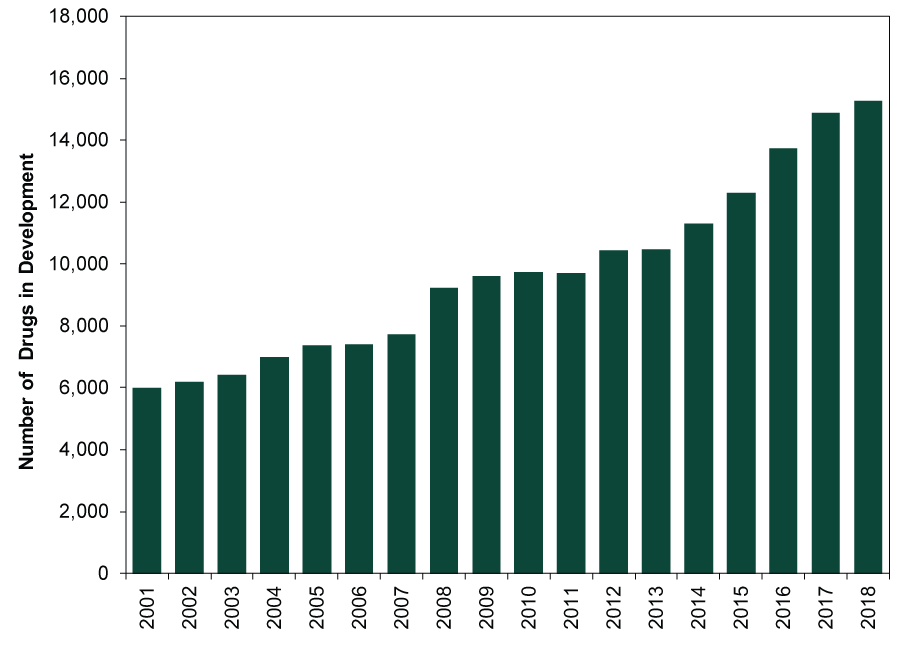
Rising drug approvals aren’t the only positive at work. Recent years have seen a drug innovation boom—particularly in specialty drugs used to treat chronic, complex or rare conditions. (Exhibit 2) From 2007 to 2017, specialty drugs’ share of global drug spending rose from 19% to 32%.[ii] Most industry analysts expect this surge to continue as medical science uncovers new opportunities to treat unmet needs.[iii] By 2022, IQVIA Institute—a clinical research organization—forecasts specialty drugs will account for more than half of projected US drug spending.[iv] Next generation cell-based and gene therapies should become key innovation drivers, with hundreds in late-stage development today. Robust drug pipelines suggest more innovations are forthcoming. (Exhibit 3)
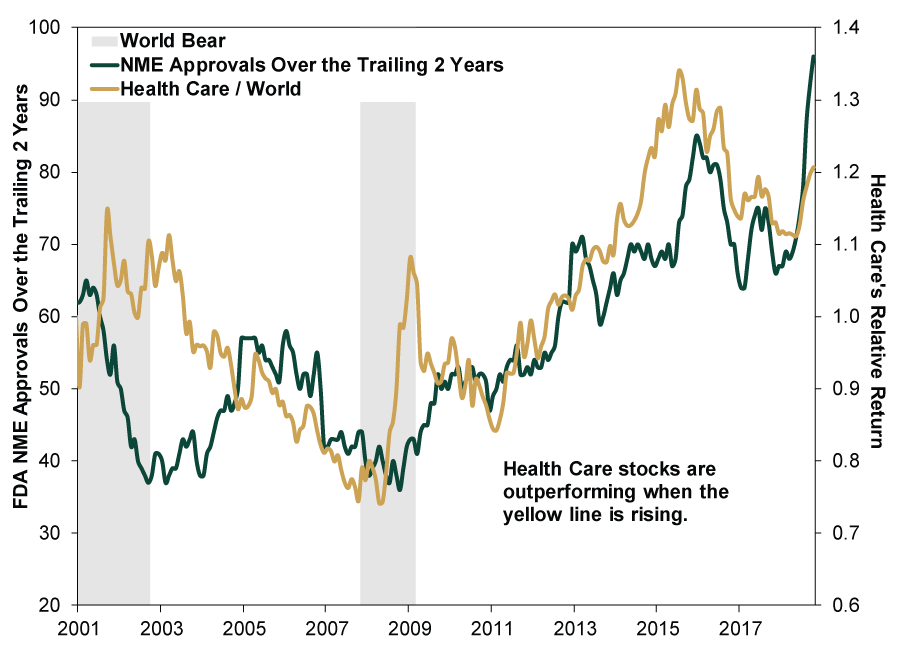
According to our research, new drug approvals are highly correlated with Health Care outperformance. This makes sense as new drugs hitting the market provide new revenue streams to help offset lost sales from drugs losing patent protection. Today, increasing innovation should help push new drugs forward. Hence, rising drug innovation and approval, alongside their size and growth orientation, should continue benefiting Heath Care stocks, in our view. (Exhibit 4)
Exhibit 2: The Third Drug Innovation Boom
Source: US Food and Drug Administration, as of 10/29/2018. New Molecular Entity (NME) approvals for the trailing five years, 1945 – 2018.
Exhibit 3: Drug Pipelines Continue to Grow
Source: Pharmaprojects, as of 10/29/2018. Total number of drugs in development, 2001 – 2018.
Exhibit 4: Health Care Outperforms in Periods of Rising Innovation
Source: FactSet, as of 10/29/2018. NME approvals and MSCI Health Care and MSCI World Index returns with net dividends, monthly, 1/31/2001 – 10/31/2018. Stock returns indexed to 1 at 1/31/2001.
If you would like to contact the editors responsible for this article, please message MarketMinder directly.
*The content contained in this article represents only the opinions and viewpoints of the Fisher Investments editorial staff.
Get a weekly roundup of our market insights
Sign up for our weekly e-mail newsletter.

You Imagine Your Future. We Help You Get There.
Are you ready to start your journey to a better financial future?

Where Might the Market Go Next?
Confidently tackle the market’s ups and downs with independent research and analysis that tells you where we think stocks are headed—and why.